 
Using the newly discovered law of quantum mechanics, the scientist postulated that: Niels Bohr, one of the giants of physics, addressed these issues in his Bohr model. 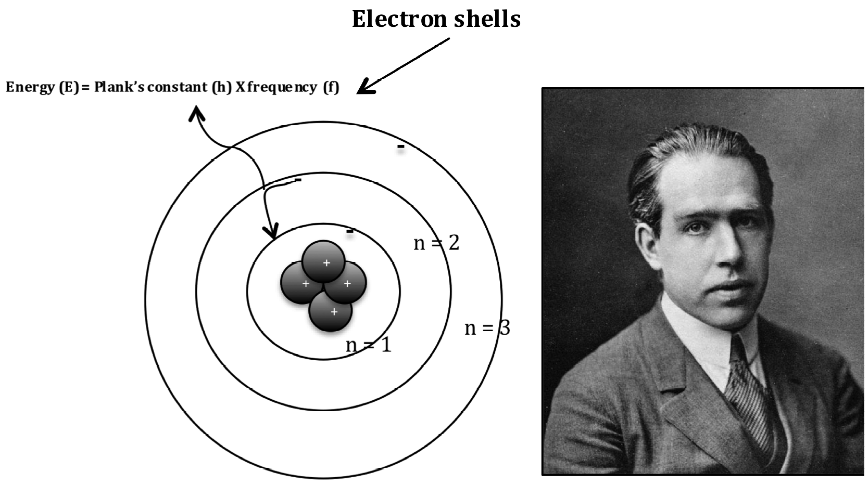
The other fundamental issue was the unexplained nature of the emission spectra of atoms. No such thing was observed, which somehow implied orbiting without energy loss. The first issue is the lack of emitted radiation: when subjected to an acceleration, a charged particle emits electromagnetic radiation at specific frequencies. The Rutherford model has a significant set of problems that kept it from describing atoms' observed behavior. In Rutherford's model, electrons orbit the atoms in orbits that resemble satellites revolving around their planets. At the beginning of the XX century, Rutherford and colleagues performed a simple experiment that proved the existence of atomic nuclei: small positively charged groups of particles (protons).Nearly a century later, Thomson introduced the newly discovered negatively charged electrons in his atomic model: with the "plum pudding mode,l" the atoms were finally divisible, but physicists were still a far shot from a realistic description of the atom.The first "modern" model for atoms was proposed by John Dalton, which postulated the "quantized" nature of matter after observing definite proportions of elements in many compounds.Bohr model is the first atomic model that introduced the principles of quantum mechanics in describing the fundamental components of matter.īefore introducing the Bohr model, let's quickly survey the previous atomic models: it will make understanding the Bohr model and its equation easier. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
